This Dog's Nose Is So Good at Smelling Cancer That Scientists Are Trying to Build One Just Like It

Claire Guest, co-founder of Medical Detection Dogs, with Daisy, whom she credits with saving her life.
Daisy wouldn't leave Claire Guest alone. Instead of joining Guest's other dogs for a run in the park, the golden retriever with the soulful eyes kept nudging Guest's chest, and stared at her intently, somehow hoping she'd get the message.
"I was incredibly lucky to be told by Daisy."
When Guest got home, she detected a tiny lump in one of her breasts. She dismissed it, but her sister, who is a family doctor, insisted she get it checked out.
That saved her life. A series of tests, including a biopsy and a mammogram, revealed the cyst was benign. But doctors discovered a tumor hidden deep inside her chest wall, an insidious malignancy that normally isn't detected until the cancer has rampaged out of control throughout the body. "My prognosis would have been very poor," says Guest, who is an animal behavioralist. "I was incredibly lucky to be told by Daisy."
Ironically, at the time, Guest was training hearing dogs for the deaf—alerting them to doorbells or phones--for a charitable foundation. But she had been working on a side project to harness dogs' exquisitely sensitive sense of smell to spot cancer at its earliest and most treatable stages. When Guest was diagnosed with cancer two decades ago, however, the use of dogs to detect diseases was in its infancy and scientific evidence was largely anecdotal.
In the years since, Guest and the British charitable foundation she co-founded with Dr. John Church in 2008, Medical Detection Dogs (MDD), has shown that dogs can be trained to detect odors that predict a looming medical crisis hours in advance, in the case of diabetes or epilepsy, as well as the presence of cancers.
In a proof of principle study published in the BMJ in 2004, they showed dogs had better than a 40 percent success rate in identifying bladder cancer, which was significantly better than random chance (14 percent). Subsequent research indicated dogs can detect odors down to parts per trillion, which is the equivalent of sniffing out a teaspoon of sugar in two Olympic size swimming pools (a million gallons).
American scientists are devising artificial noses that mimic dogs' sense of smell, so these potentially life-saving diagnostic tools are widely available.
But the problem is "dogs can't be scaled up"—it costs upwards of $25,000 to train them—"and you can't keep a trained dog in every oncology practice," says Guest.
The good news is that the pivotal 2004 BMJ paper caught the attention of two American scientists—Andreas Mershin, a physicist at MIT, and Wen-Yee Yee, a chemistry professor at The University of Texas at El Paso. They have joined Guest's quest to leverage canines' highly attuned olfactory systems and devise artificial noses that mimic dogs' sense of smell, so these potentially life-saving diagnostic tools are widely available.
"What we do know is that this is real," says Guest. "Anything that can improve diagnosis of cancer is something we ought to know about."
Dogs have routinely been used for centuries as trackers for hunting and more recently, for ferreting out bombs and bodies. Dogs like Daisy, who went on to become a star performer in Guest's pack of highly trained cancer detecting canines before her death in 2018, have shared a special bond with their human companions for thousands of years. But their vastly superior olfaction is the result of simple anatomy.
Humans possess about six million olfactory receptors—the antenna-like structures inside cell membranes in our nose that latch on to the molecules in the air when we inhale. In contrast, dogs have about 300 million of them and the brain region that analyzes smells is, proportionally, about 40 times greater than ours.
Research indicates that cancerous cells interfere with normal metabolic processes, prompting them to produce volatile organic compounds (VOCs), which enter the blood stream and are either exhaled in our breath or excreted in urine. Dogs can identify these VOCs in urine samples at the tiniest concentrations, 0.001 parts per million, and can be trained to identify the specific "odor fingerprint" of different cancers, although teaching them how to distinguish these signals from background odors is far more complicated than training them to detect drugs or explosives.
For the past fifteen years, Andreas Mershin of MIT has been grappling with this complexity in his quest to devise an artificial nose, which he calls the Nano-Nose, first as a military tool to spot land mines and IEDS, and more recently as a cancer detection tool that can be used in doctors' offices. The ultimate goal is to create an easy-to-use olfaction system powered by artificial intelligence that can fit inside of smartphones and can replicate dogs' ability to sniff out early signs of prostate cancer, which could eliminate a lot of painful and costly biopsies.
Trained canines have a better than 90 percent accuracy in spotting prostate cancer, which is normally difficult to detect. The current diagnostic, the prostate specific antigen test, which measures levels of certain immune system cells associated with prostate cancer, has about as much accuracy "as a coin toss," according to the scientist who discovered PSA. These false positives can lead to unnecessary and horrifically invasive biopsies to retrieve tissue samples.
So far, Mershin's prototype device has the same sensitivity as the dogs—and can detect odors at parts per trillion—but it still can't distinguish that cancer smell in individual human patients the way a dog can. "What we're trying to understand from the dogs is how they look at the data they are collecting so we can copy it," says Mershin. "We still have to make it intelligent enough to know what it is looking at—what we are lacking is artificial dog intelligence."

The intricate parts of the artificial nose are designed to fit inside a smartphone.
At UT El Paso, Wen-Yee Lee and her research team has used the canine olfactory system as a model for a new screening test for prostate cancer, which has a 92 percent accuracy in tests of urine samples and could be eventually developed as a kit similar to the home pregnancy test. "If dogs can do it, we can do it better," says Lee, whose husband was diagnosed with prostate cancer in 2005.
The UT scientists used samples from about 150 patients, and looked at about 9,000 compounds before they were able to zero in on the key VOCs that are released by prostate cancers—"it was like finding a needle in the haystack," says Lee. But a more reliable test that can also distinguish which cancers are more aggressive could help patients decide their best treatment options and avoid invasive procedures that can render them incontinent and impotent.
"This is much more accurate than the PSA—we were able to see a very distinct difference between people with prostate cancer and those without cancer," says Lee, who has been sharing her research with Guest and hopes to have the test on the market within the next few years.
In the meantime, Guest's foundation has drawn the approving attention of royal animal lovers: Camilla, the Duchess of Cornwall, is a patron, which opened up the charitable floodgates and helped legitimize MDD in the scientific community. Even Camilla's mother-in-law, Queen Elizabeth, has had a demonstration of these canny canines' unique abilities.

Claire Guest, and two of MDDs medical detection dogs, Jodie and Nimbus, meet with queen Elizabeth.
"She actually held one of my [artificial] noses in her hand and asked really good questions, including things we hadn't thought of, like the range of how far away a dog can pick up the scent or if this can be used to screen for malaria," says Mershin. "I was floored by this curious 93-year-old lady. Half of humanity's deaths are from chronic diseases and what the dogs are showing is a whole new way of understanding holistic diseases of the system."
Thanks to safety cautions from the COVID-19 pandemic, a strain of influenza has been completely eliminated.
If you were one of the millions who masked up, washed your hands thoroughly and socially distanced, pat yourself on the back—you may have helped change the course of human history.
Scientists say that thanks to these safety precautions, which were introduced in early 2020 as a way to stop transmission of the novel COVID-19 virus, a strain of influenza has been completely eliminated. This marks the first time in human history that a virus has been wiped out through non-pharmaceutical interventions, such as vaccines.
The flu shot, explained
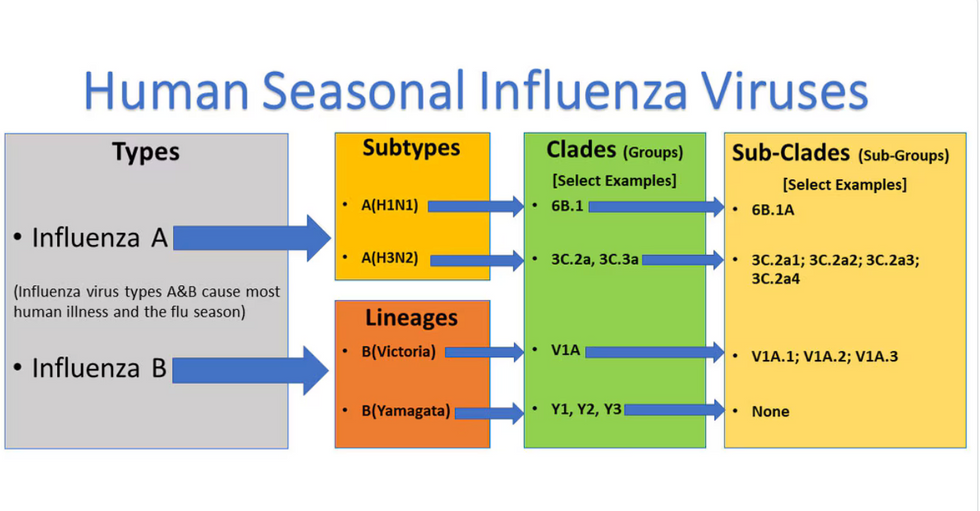
Influenza viruses type A and B are responsible for the majority of human illnesses and the flu season.
Centers for Disease Control
For more than a decade, flu shots have protected against two types of the influenza virus–type A and type B. While there are four different strains of influenza in existence (A, B, C, and D), only strains A, B, and C are capable of infecting humans, and only A and B cause pandemics. In other words, if you catch the flu during flu season, you’re most likely sick with flu type A or B.
Flu vaccines contain inactivated—or dead—influenza virus. These inactivated viruses can’t cause sickness in humans, but when administered as part of a vaccine, they teach a person’s immune system to recognize and kill those viruses when they’re encountered in the wild.
Each spring, a panel of experts gives a recommendation to the US Food and Drug Administration on which strains of each flu type to include in that year’s flu vaccine, depending on what surveillance data says is circulating and what they believe is likely to cause the most illness during the upcoming flu season. For the past decade, Americans have had access to vaccines that provide protection against two strains of influenza A and two lineages of influenza B, known as the Victoria lineage and the Yamagata lineage. But this year, the seasonal flu shot won’t include the Yamagata strain, because the Yamagata strain is no longer circulating among humans.
How Yamagata Disappeared

Flu surveillance data from the Global Initiative on Sharing All Influenza Data (GISAID) shows that the Yamagata lineage of flu type B has not been sequenced since April 2020.
Nature
Experts believe that the Yamagata lineage had already been in decline before the pandemic hit, likely because the strain was naturally less capable of infecting large numbers of people compared to the other strains. When the COVID-19 pandemic hit, the resulting safety precautions such as social distancing, isolating, hand-washing, and masking were enough to drive the virus into extinction completely.
Because the strain hasn’t been circulating since 2020, the FDA elected to remove the Yamagata strain from the seasonal flu vaccine. This will mark the first time since 2012 that the annual flu shot will be trivalent (three-component) rather than quadrivalent (four-component).
Should I still get the flu shot?
The flu shot will protect against fewer strains this year—but that doesn’t mean we should skip it. Influenza places a substantial health burden on the United States every year, responsible for hundreds of thousands of hospitalizations and tens of thousands of deaths. The flu shot has been shown to prevent millions of illnesses each year (more than six million during the 2022-2023 season). And while it’s still possible to catch the flu after getting the flu shot, studies show that people are far less likely to be hospitalized or die when they’re vaccinated.
Another unexpected benefit of dropping the Yamagata strain from the seasonal vaccine? This will possibly make production of the flu vaccine faster, and enable manufacturers to make more vaccines, helping countries who have a flu vaccine shortage and potentially saving millions more lives.
After his grandmother’s dementia diagnosis, one man invented a snack to keep her healthy and hydrated.
Founder Lewis Hornby and his grandmother Pat, sampling Jelly Drops—an edible gummy containing water and life-saving electrolytes.
On a visit to his grandmother’s nursing home in 2016, college student Lewis Hornby made a shocking discovery: Dehydration is a common (and dangerous) problem among seniors—especially those that are diagnosed with dementia.
Hornby’s grandmother, Pat, had always had difficulty keeping up her water intake as she got older, a common issue with seniors. As we age, our body composition changes, and we naturally hold less water than younger adults or children, so it’s easier to become dehydrated quickly if those fluids aren’t replenished. What’s more, our thirst signals diminish naturally as we age as well—meaning our body is not as good as it once was in letting us know that we need to rehydrate. This often creates a perfect storm that commonly leads to dehydration. In Pat’s case, her dehydration was so severe she nearly died.
When Lewis Hornby visited his grandmother at her nursing home afterward, he learned that dehydration especially affects people with dementia, as they often don’t feel thirst cues at all, or may not recognize how to use cups correctly. But while dementia patients often don’t remember to drink water, it seemed to Hornby that they had less problem remembering to eat, particularly candy.

Hornby wanted to create a solution for elderly people who struggled keeping their fluid intake up. He spent the next eighteen months researching and designing a solution and securing funding for his project. In 2019, Hornby won a sizable grant from the Alzheimer’s Society, a UK-based care and research charity for people with dementia and their caregivers. Together, through the charity’s Accelerator Program, they created a bite-sized, sugar-free, edible jelly drop that looked and tasted like candy. The candy, called Jelly Drops, contained 95% water and electrolytes—important minerals that are often lost during dehydration. The final product launched in 2020—and was an immediate success. The drops were able to provide extra hydration to the elderly, as well as help keep dementia patients safe, since dehydration commonly leads to confusion, hospitalization, and sometimes even death.
Not only did Jelly Drops quickly become a favorite snack among dementia patients in the UK, but they were able to provide an additional boost of hydration to hospital workers during the pandemic. In NHS coronavirus hospital wards, patients infected with the virus were regularly given Jelly Drops to keep their fluid levels normal—and staff members snacked on them as well, since long shifts and personal protective equipment (PPE) they were required to wear often left them feeling parched.
In April 2022, Jelly Drops launched in the United States. The company continues to donate 1% of its profits to help fund Alzheimer’s research.


